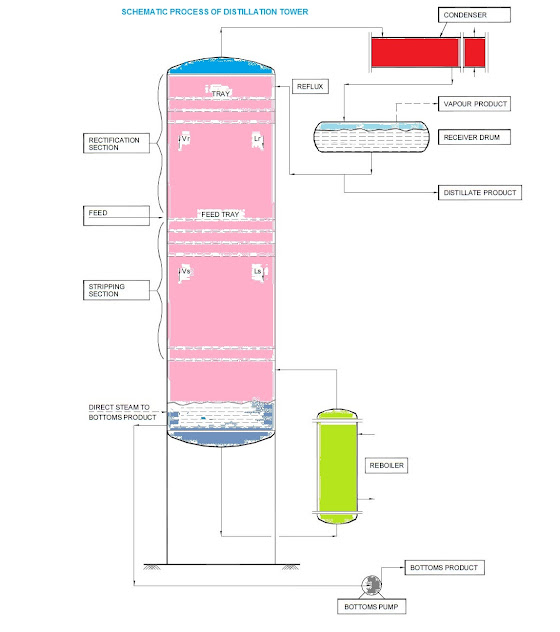
Distillation: The distillation is separation of the constituents of a liquid mixture via partial vaporization of the mixture and separate recovery of vapour and residue. Various kinds of devices called plates or trays are used to bring the two phases into intimate contact. The trays are stacked one above the other and enclosed in a cylindrical shell to form a column. Distillation is very important process.
The feed material, which is to be separated into fractions, is introduced at one or more points along the column shell. Due to difference in gravity between liquid and vapour phases, the liquid runs down the column, cascading from tray to tray, while vapour goes up the column contacting the liquid at each tray.
Distillation
The liquid reaching the bottom of the column is partially vaporised in a heated reboiler to provide reboil vapour, which is sent back up the column. The remainder of the bottom liquid is withdrawn as the bottom product. The vapour reaching the top of column is cooled and condensed to a liquid in the overhead condenser.
Part of this liquid is returned to the column as reflux to provide liquid overflow and to control the temperature of the fluids in the upper portion of the tower. The remainder of the overhead stream is withdrawn as the overhead or distillate product.
Absorption and Stripping
Many operations in petrochemical plants require the absorption of components from gas streams into lean oils or solvents. The resultant rich oil is then stripped or denuded of the absorbed materials.
The greatest use of this operation utilises hydrocarbon materials, but the principles are applicable to other systems provided adequate equilibrium data is available.
Fractionation
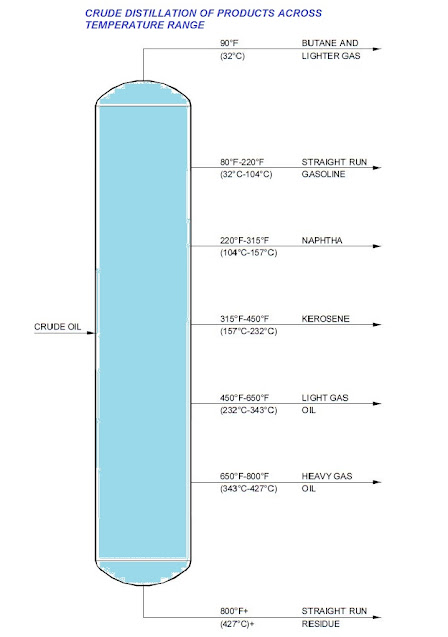
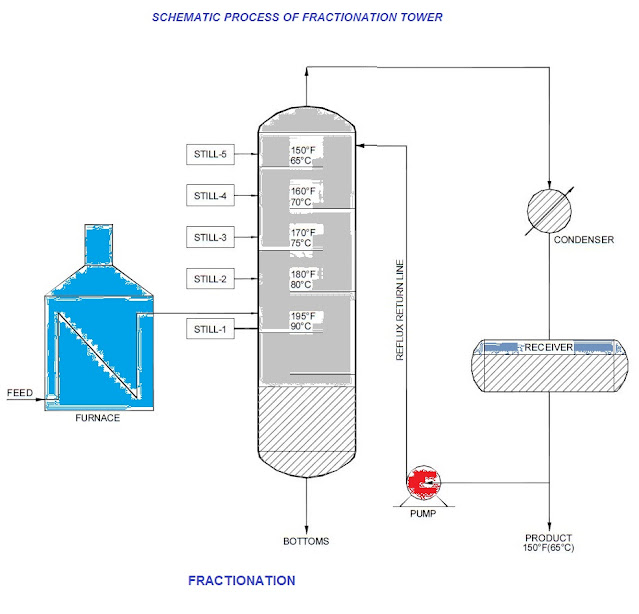
A fractionation column is a type of still. A simple still starts with mixed liquids, such as alcohol and water produced by fermenting grain etc. and by boiling produces a distillate in which the concentration of alcohol is many times higher than in feed. In petroleum industry, mixtures of not only two but a lot many components are dealt with.
Crude oil is a typical feed for a fractionation column and from it, the column can form simultaneously several distillates such as wax distillate, gas oil, heating oil, naptha and fuel gas. These fractions are termed cuts. The feed is heated in a furnace before it enters the column. As the feed enters the column, quantities of vapour are given off by flashing due to release of pressure on the feed.
As the vapours rise up the column, they come into intimate contact with down flowing liquid. During this contact, some of the heavier components of the vapour are condensed and some of the higher components of the down flowing liquid are vaporised.
This process is termed refluxing. If the composition of the feed remains the same and the column is kept in steady operation, a temperature distribution establishes in the column. The temperature at any tray is the boiling point of the liquid on the tray. ‘Cuts’ are not taken from every tray.
The P&ID will show cuts that are to be made, including alternatives. Nozzles on selected trays are piped and nozzles for alternate operation are provided with line blinds or valves.
Stripper is used to strip lighter materials from bottom of a main or a vacuum tower distilling crude bottom residue under vacuum.



1 thought on “what is distillation, absorption and stripping, fractionation column.”