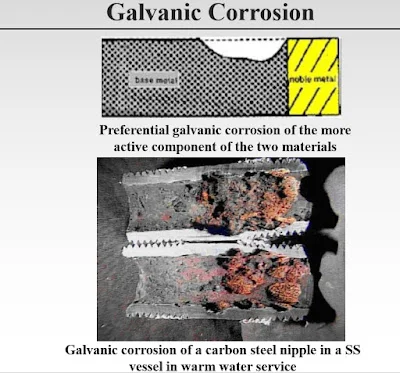
Galvanic corrosion may occur when two different metallic materials are connected in corrosive service. The potential difference may cause an increased corrosion rate for the less noble material.
If neither of the two materials corrodes by itself in a service, combination with the other will not normally cause corrosion in this service.
In this aspect, copper-based materials are not considered corrosion resistant in untreated seawater and shall always be isolated from more corrosion resistant materials like 25% Cr duplex or titanium.
For submerged galvanic couples, the cathodic protection system will prevent galvanic corrosion.
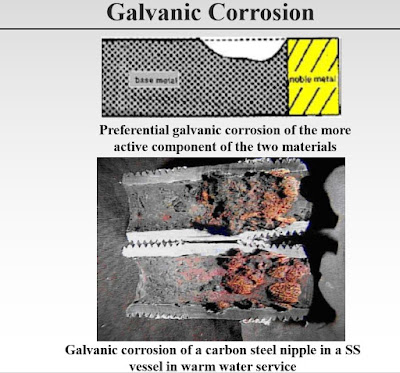
At connections between dissimilar materials without an isolation/distance spool, it can be assumed that the local corrosion rate near the interface is approximately three times higher than the average corrosion rate, decreasing exponentially away from the interface within a length of ten pipe diameters.
This should be used to establish the magnitude of the corrosion allowances. Particular systems may have higher corrosion rates depending on area ratio, material combinations, and the corrosion environment.
If two materials are connected in corrosive service, galvanic corrosion shall be mitigated by one of the following methods:
- Internally coating the more noble material (e.g., stainless steel) from the junction with the less noble material (e.g., carbon steel) to approximately ten pipe diameters. It shall be verified that the coating is compatible with the fluid.
- Use a thick-walled carbon steel flanged section at the junction, which is designed for replacement at scheduled intervals
- Use a spool piece of a non-conducting material (e.g., FRP or rubber lined carbon steel) with a length of about ten times the pipe diameter.
- Install internal sacrificial anodes through access fittings near the interface. This works only when the system is filled with a conductive liquid, and service conditions are compatible with the anodes. Special precautions during commissioning and shut-in are required. Process engineers shall confirm that there will not be any negative effect on the process by corrosion product from the anodes nor will process upsets cause excessive consumption rates for the anodes.
- Insulating gaskets are not effective in preventing galvanic corrosion and shall not be used.

1 thought on “what is Galvanic corrosion”